Funding
2021 Spinout funding - Innovate UK and UCSF
HydRegen has been awarded funding to form a company and spin out from the University of Oxford. The Company was incorporated in Dec 2020 and expects to start R&D in March 2021.
2020 ICURe Scheme - Holly Reeve is Entrepreneurial Lead
The HydRegen pre-spin out has been awarded a place on the
ICURe scheme to carry out market validation research. This is led by Dr Holly Reeve.
2020 BIA PULSE programme - Holly Reeve selected
Holly was selected for the intensive
BIA PULSE leadership and entreprenuership training programme developed by the BIA and the Francis Crick Institute, aimed at CEOs and future CEOs of companies in the life sciences.
2019 iCASE studentship
Charlotte Hancox joined the team to begin her DPhil; her research project is funded by a BBSRC iCASE Studentship through the Interdisciplinary Bioscience DTP, with support from
Johnson Matthey.
2017 Public Engagement with Research Funding
Holly has been awarded
funding by the MPLS division to support public engagement activties . We will develop a series of games, talks and experiments to take to secondary schools and build an online platform to inspire and inform school students about Industrial Biotechnology and the
HydRegen technology.
2017 iCASE Studentship
Barnabas Poznansky joined the team to begin his DPhil; his research project looks at operating the enzyme-modified particle system in continuous flow. This is funded by an iCASE studentship through the BBSRC Interdisciplinary Bioscience DTP, with support from
Dr Reddy's.
2016 IB Catalyst funding
The Vincent group have received major funding from EPSRC via Innovate UK / EPSRC / BBSRC Industrial Biotechnology Catalyst Round 3 (
EP/N013514/1). The project, developing H
2-driven enzyme-catalysed chemical synthesis, started in Jan 2016 with Dr Holly Reeve as Project Manager.
2015 iCASE studentship
Michalis Posidias joined the team to begin his DPhil, his research project looks at operating the enzyme-modified particle system in the reverse direction for H
+-driven NAD
+ recycling to support terminal alcohol oxidation to generate aldehydes. This is funded by a BBSRC iCASE studentship with support from
Johnson Matthey Catalysis and Chiral Technologies.
2014 Business Interaction Voucher
Work to extend the H
2-driven cofactor recycling system to NADPH recycling for NADPH-dependent enzymes was supported by a BBSRC Metals in Biology NIBB Business Interaction Voucher with GSK as collaborator.
2012 ERC Proof of Concept funding
R&D on this innovation was supported by ERC Proof of Concept Grant 297503 during 2012.
Publications
Front Cover Article in ACS Catalysis: J.S. Rowbotham, H.A. Reeve, K.A. Vincent, 'Hybrid Chemo-, Bio-, and Electrocatalysis for Atom-Efficient Deuteration of Cofactors in Heavy Water',
ACS Catalysis, 2021, 11, 2596-2604,
DOI: 110.1021/acscatal.0c03437
J.S. Rowbotham, A.P. Hardy, H.A. Reeve, K.A. Vincent, 'Synthesis of [4S-
2H]NADH, [4R-
2H]NADH, [4-
2H
2]NADH and [4-
2H]NAD
+ cofactors through heterogeneous biocatalysis in heavy water',
J Label Compd Radiopharm., 2021, 1-6,
DOI: 10.1002/jlcr.3899
Preprint: Zhao, X., Cleary, S.E., Zor, C., Grobert, N., Reeve, H.A., Vincent, K.A., 'Chemo-Bio Catalysis Using Carbon Supports: Application in H
2-Driven Cofactor Recycling',
ChemRxiv, 2020,
Link
Preprint: Poznansky, B., Thompson, L.A., Reeve, H.A., Vincent, K.A., 'Accelerating Biocatalytic Hydrogenations using the H-Cube Flow Reactor',
ChemRxiv, 2020,
Link
Preprint: Srinivasan, S.J., Cleary, S.E., Paul, E.P., Ramirez, M.A., Vincent, K.A., '
E. coli Nickel-Iron Hydrogenase 1 Catalyses Non-native
Reduction of Flavins: Demonstration for Alkene Hydrogenation by Old Yellow Enzyme',
ChemRxiv,
Link
Preissler, J., Reeve, H.A., Zhu, T., Nicholson, J., Urata, K., Lauterbach, L., Wong, L.L., Vincent, K.A., Lenz, O., 'Dihydrogen-driven NADPH recycling in imine reduction and P450-catalyzed oxidations mediated by an engineered O
2-tolerant hydrogenase',
ChemCatChem, 2020,
12, 4835-4851,
DOI: 10.1002/cctc.202000763
Thompson, L.A., Rowbotham, J.S., Nicholson, J.H., Ramirez, M.A., Zor, C., Reeve, H.A., Grobert, N., Vincent, K.A., 'Rapid, Heterogeneous Biocatalytic Hydrogenation and Deuteration in a Continuous Flow Reactor',
ChemCatChem, 2020,
12, 3913-3918,
DOI: 10.1002/cctc.202000161
J.S. Rowbotham, M.A. Ramirez, O. Lenz, H.A. Reeve, K.A. Vincent, 'Bringing biocatalytic deuteration into the toolbox of asymmetric isotopic labelling techniques',
Nature Communications, 2020,
11, 1454,
DOI: 10.1038/s41467-020-15310-z
B. Poznansky, L.A. Thompson, S.A. Warren, H.A. Reeve, K.A. Vincent, 'Carbon as a Simple Support for Redox Biocatalysis in Continuous Flow',
Org. Process Res. Dev., 2020, Special Issure:
Flow Chemistry Enabling Efficient Synthesis.
DOI: 10.1021/acs.oprd.9b00410
Thompson, L.A., Rowbotham, J.S., Reeve, H.A., Zor, Z., Grobert, N., Vincent, K.A., Chapter 18: Biocatalytic Hydrogenations on Carbon Supports in 'Nanoarchitectures Built with Carbon Nanotubes and Magnetic Nanoparticles',
Methods in Enzymology, Editor - Kumar, 2020, Vol. 630, pp303-325.
DOI: 10.1016/bs.mie.2019.10.017
Preprint: Rowbotham, J.S., Lenz, O., Reeve, H.A., Vincent, K.A., 2019,
ChemRxiv DOI: https://doi.org/10.26434/chemrxiv.7982864.v1
Cover article in ChemCommun: Zor, C., Reeve, H.A., Quinson, J., Thompson, L.A., Lonsdale, T.H., Dillon, F., Grobert, N., Vincent K.A., 'H
2-driven Biocatalytic Hydrogenation in Continuous Flow using Enzyme-Modified Carbon Nanotube Columns',
Chem. Commun., 2017, 53, 9839-9841
DOI:10.1039/C7CC04465H
Cover Article in Biochemical Journal: Reeve, H.A., Ash, P.A., Park, H., Huang, A., Posidias, M., Tomlinson, C., Lenz, O., Vincent, K.A., 'Enzymes as modular catalysts for redox half reactions in H
2-powered chemical synthesis: from biology to technology',
Biochemical Journal, 2017,
474, 215-230
DOI:10.1042/BCJ20160513
Cover Article in ChemCatChem:
Reeve, H.A., Lauterbach, L., Lenz, O., Vincent, K.A. 'Enzyme-Modified Particles for Selective Bio-Catalytic Hydrogenation via H
2-driven NADH Recycling'
ChemCatChem, 2015,
7, 21, 3480 - 3487
DOI:10.1002/cctc.201500766
Reeve, H.A., Lauterbach, L., Ash, P.A., Lenz, O., Vincent, K.A., 'A modular
system for regeneration of NAD cofactors using graphite particles modified with
hydrogenase and diaphorase moieties'
Chem. Commun. 2012, 48 (10), 1589-1591.
Link.
Lauterbach, L., Idris, Z., Vincent, K.A., Lenz, O. 'Catalytic properties of the
isolated diaphorase fragment of the NAD
+-reducing [NiFe]-hydrogenase
from
Ralstonia eutropha'
PLoS ONE, 2011, 6, (10): e25939.
Link.
Intellectual Property
A patent covering the HydRegen technology was filed in 2011 and is granted in the EU and in the final stages of consideration in the US.
Publication number:
WO2013050760 A2.
More information about the technology can be found on the
Oxford University Innovation website.
Two further patent applications have been filed through OUI, with further IP protection in progress.
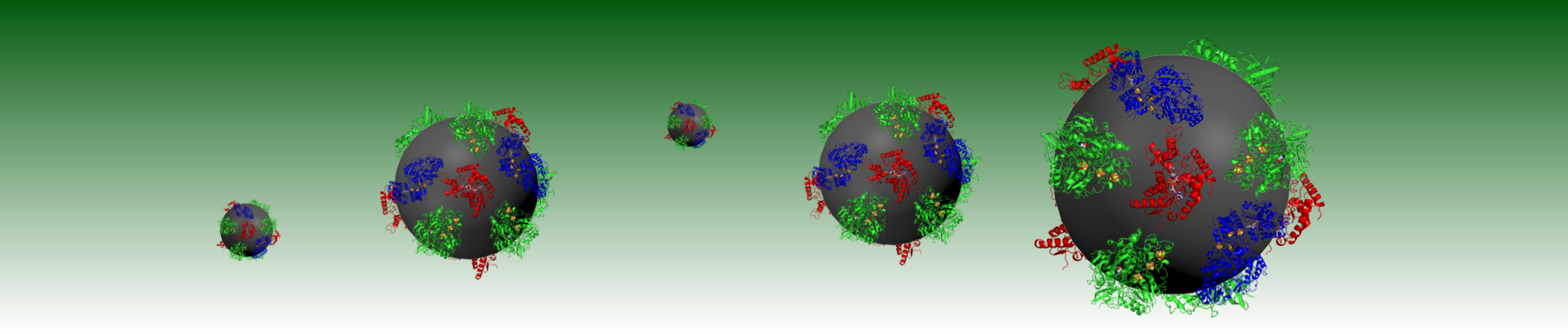




 The HydRegen Technology was the Overall Winner at the RSC Emerging Technology Competition on the 5th June 2013.
The HydRegen Technology was the Overall Winner at the RSC Emerging Technology Competition on the 5th June 2013.

